What is Ultraviolet Radiation?
What is Ultraviolet Radiation?
Ultraviolet radiation, also called ultraviolet light, is a type of electromagnetic radiation on the same spectrum as visible light. Ultraviolet is frequently abbreviated as “UV”. UV radiation is very energetic compared to visible light or infrared radiation and is strong enough to destroy some chemical bonds when it collides with molecules. It is emitted by the sun as part of the solar radiation spectrum and has consequences for people and objects on the surface of the Earth.
The wavelength of any type of electromagnetic radiation is defined as the size of the wave that the energy is composed of, and it is measured in nanometers, which is one billionth of a meter. As a part of the electromagnetic spectrum, UV is just beneath the visible portion of the spectrum and is between 100 and 380 nanometers long. Additionally, the ultraviolet portion of the electromagnetic radiation is further broken down into three bands, called UVA, UVB, and UVC. UVA is the longest band, from 320 to 380 nanometers, and goes through glass unimpeded. UVB radiation is more energetic, ranging from 280 to 320 12.4eV, and this energy value is a measure that can be used to predict the effect it will have on substances.
The Destructive Energy of UV
Because ultraviolet radiation is very energetic, it is also very destructive. A useful analogy is to compare dropping a grape and a steel ball bearing onto a sheet of paper. A photon of visible light is like dropping a grape onto the surface of a suspended piece of paper, where the grape will stop at the surface or bounce off of the paper. A photon of ultraviolet light, however, is like dropping a steel ball bearing onto the surface of a suspended piece of paper. The steel bearing may also stop at the surface of the paper, though there is also a chance that it will punch a hole in the paper and drop through. Thus, when UV radiation collides with some substances, the energy of the interaction is strong enough to shatter chemical bonds and destroy molecules. The process of a photon striking a molecule and breaking it apart is called “photodissociation”.

UV radiation will sometimes destroy molecules in ways that affect human bodies and environments in undesirable ways. The energy (eV) of a UV photon determines its ability to shatter chemical bonds when it impacts them, which is why the shorter the wavelength of UV, the more destructive it is. UVA photons are very energetic, and they have a strong ability to cause photodissociation. There are numerous molecules with chemical bonds weak enough for UVA to destroy, such as skin or fabrics. UVB is even more energetic than UVA and has the ability to photodissociate even more molecules because it is capable of destroying even stronger bonds. UVC is the most energetic band of ultraviolet radiation, and it is violently destructive; though the ozone layer greatly limits the ability of UVC from reaching the Earth’s surface. UVC is often used in laboratories to kill bacteria and viruses.
UVA and Skin
Although UVA is the least destructive band of ultraviolet radiation, it still contributes significantly to premature aging and fading. According to the University of Maryland Medical Center, UVA and UVB together account for 90% of all premature skin aging and wrinkles (1). Additionally, UVA is more efficient at penetrating the skin than UVB or UVC, and it penetrates deeper as well. As a result, UVA exposure is a problem year round. One cause of skin damage from UVA is that it destroys vitamin A in the skin, which contributes to cell damage. Collagen, the structural protein that keeps skin cells and other cells firm, is also damaged by exposure to UVA radiation. Prolonged damage to this structural protein, which occurs at an energy of 3.5eV, causes the skin to wrinkle and sag over time, and is the source of “sun wrinkles.” Additionally, UVA radiation is also a risk factor for skin cancer and is increasingly being linked to the disease.
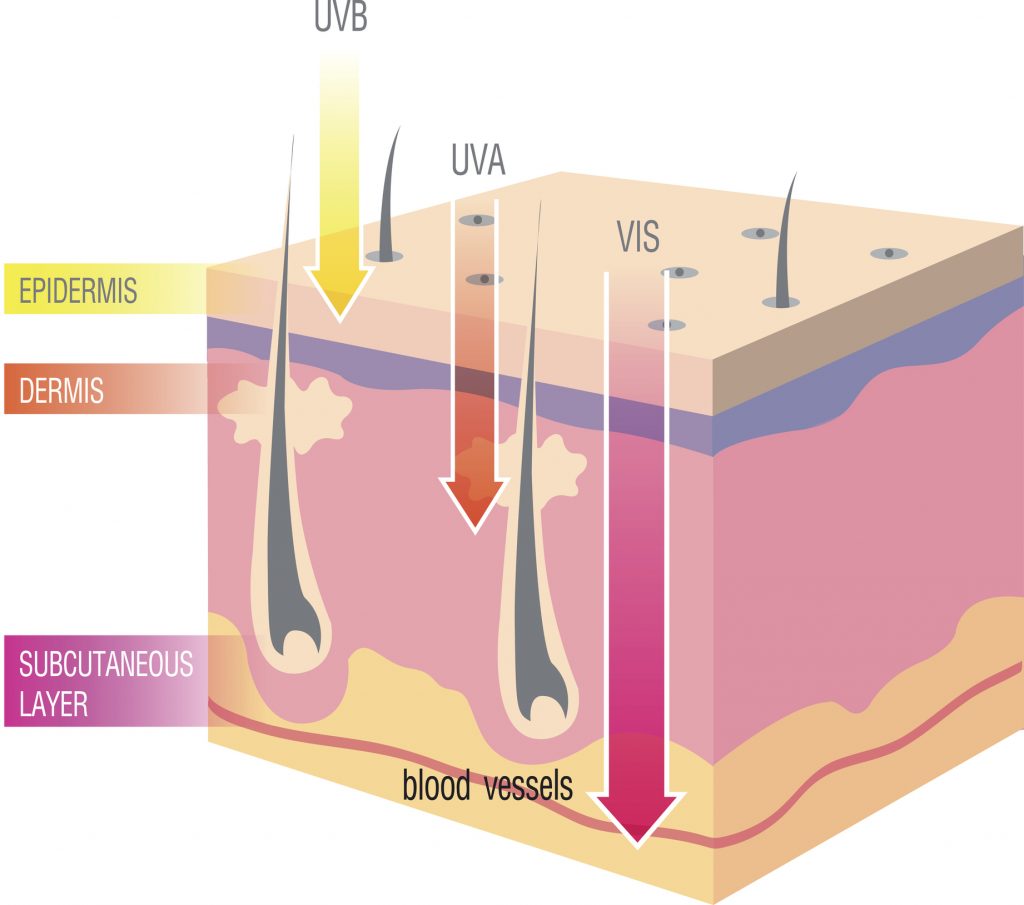
Skin is not the only organ affected by UVA, as this band of ultraviolet radiation is also implicated in causing cataracts of the eye and damage to the macula, a part of the eye that processes central vision. UVA exposure is also associated with immunosuppression, a reduction in the ability of the immune system to fight off disease (2, 3).
UVB and Skin
UVB is also a major contributor to premature aging, but the effects on human skin to exposure at this band become even more dramatic because the UVB photon energies of 3.9 to 4.4 eV are energetic enough to penetrate skin cells and shatter even more molecules within them. As a result, prolonged exposure to UVB may result in sunburn, particularly for people with lighter skin.
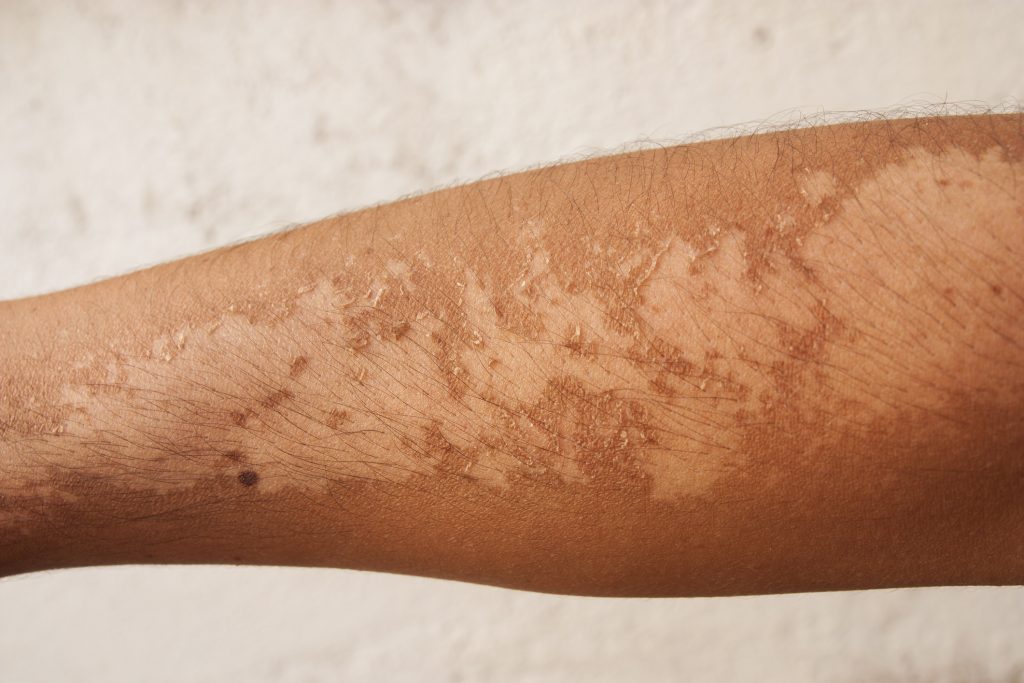
Another catastrophic result of the alteration of molecules by UVB is the creation of “free radicals” in the skin. Free radicals are very reactive molecules or ions that can damage cells and DNA. While direct damage to DNA from UVB radiation is common, most UVB damage to DNA is actually caused by the radiation breaking apart other molecules in a cell, which then become free radicals, and have the ability to react with and damage DNA. The result is often melanoma and squamous cell carcinoma cancers. Public awareness of the carcinogenic effects of free radicals is increasing, as is awareness of “antioxidants”, which reverse these chemical reactions in the body. However, there is no evidence that foods high in antioxidants will reduce the risk of skin cancer from exposure to UV radiation. UVB is also a risk factor for the eyes just as UVA is, and also contributes to cataracts and macular damage.
Fading and Decomposition
The active mechanism that causes fading and decomposition of furnishings and other property is the same mechanism that causes direct DNA mutations that result in skin cancer. UV photons, which have very high energy, can destroy some molecular bonds when they collide with them. When such a high energy photon collides with a dye molecule, the dye molecule may photodissociate and will no longer provide the pigmentation that provides the object with its color, resulting in fading. Additionally, some polymers (plastics) can undergo UV degradation due to photodissociation, contributing to the structural damage of an object.
The fading of furnishings and property is attributed in part to UVA, which has enough energy at 3.3 to 3.9 eV to contribute significantly to fading. The more energetic UVB band, with energies between 3.9 to 4.4 eV, causes fading even faster than UVA and affects more materials. UVC is highly destructive to objects and property, but again, the ozone layer blocks most UVC in the upper atmosphere.
Window Film and UV
UVC is very energetic radiation and is extremely destructive, however, people on the surface of the Earth are protected from these harmful rays by the Earth’s ozone layer. Ozone is a gas in the upper atmosphere that absorbs UVC radiation and prevents it from reaching the surface of the Earth. In this respect, adding a protective solar control film to windows is like adding a “UVA and UVB ozone layer” between the sun and the interior your home or automobile. The absorbers secured in the window film act just like ozone and absorb UVA and UVB, which provides a solid line of defense against ultraviolet radiation. While glass alone blocks a significant portion of UVB
Solar Gard window film will stop the rest of it, along with UVA, blocking over 99% of all UV of any type.
Untreated glass has the ability to block a significant portion of UVB radiation. However, unfilmed windows made of glass, even specialty glass such as low emissivity or tinted panes, are not as effective at blocking ultraviolet radiation as solar control window film. Some glass and window manufacturers provide glass with low emissivity coatings, solar control coatings, or other functional treatments that do reduce the amount of UV transmitted into a home or auto. Though some of these products block up to 95% of UV radiation, only applied window films such as Solar Gard are capable of eliminating over 99% of solar UV. A typical low-emissivity coating only blocks about 30% of the UV incident on the window. The UV absorbers in window film are suspended in the adhesives and the polyester film itself, which are more capable of blocking UV than coated glass. Therefore, solar control window film closes the transmission gaps left by the high-performance glass, achieving the highest level of UV protection.

Because window film blocks over 99% of all ultraviolet radiation, the solar protection factor (SPF) of window film
is extremely high, especially in comparison to commercial sunscreen lotions. Most people are familiar with the SPF value of sunscreen. Applying such a solar control film to the windows of one’s home or auto is like shielding the interior, protecting the inhabitants and objects within with an SPF of hundreds or thousands.
As a final note on solar control window film and the preservation of skin and health, some products, such as the Panorama brand of designer films, are recommended by the American Skin Cancer Foundation for preserving skin health. Additionally, the reduction in exposure to UV radiation of any type is recommended for people with many health conditions that can affect the skin, such as amelanism or lupus. People at risk should consult with their physician to decide if installing UV blocking film on their home and automobile is a recommended protection.



